New COVID Preprint out from BioBE:
Preprint Link: https://www.researchsquare.com/article/rs-940891/v1
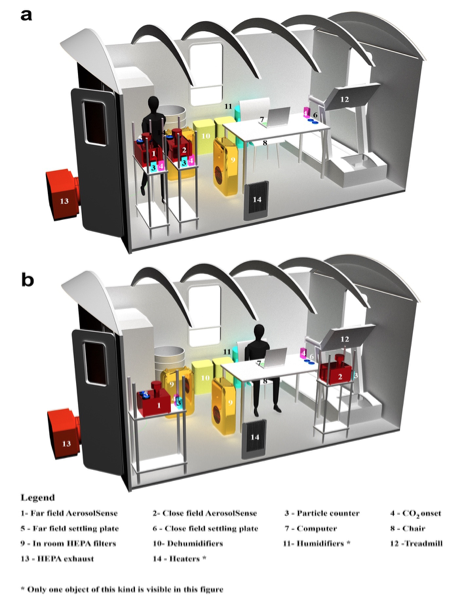
We recruited 11 participants that were diagnosed with COVID-19 and studied each for 3 days in a controlled chamber with multiple independent variables. We collected more than 2500 biological samples, particulates, and CO2 in near field (1.2 m) and far field (3.5 m) per each trial.
Each participant conducted a cough test, speak test, and speak loudly test at the beginning of each day, followed by routine trials where participants conducted regular office activities under different outdoor air exchange rates, in-room filtration, and relative humidity levels.
Below is the abstract. Further down is a summary in a brief question and answer format. Below that is a list of links to recent relevant BioBE COVID research. Enjoy the preprint and please share your comments!
Abstract: Several studies indicate that COVID-19 is primarily transmitted within indoor spaces. Therefore, environmental characterization of SARS-CoV-2 viral load with respect to human activity, building parameters, and environmental mitigation strategies is critical to combat disease transmission. We recruited 11 participants diagnosed with COVID-19 to individually occupy a controlled chamber and conduct specified physical activities under a range of environmental conditions; we collected human and environmental samples over a period of three days for each participant. Here we show that increased viral load, measured by lower RNA cycle threshold (CT) values, in nasal samples is associated with higher viral loads in environmental aerosols and surfaces captured in both the near field (1.2 m) and far field (3.5 m). At ambient conditions with ~0 Air Changes per Hour (ACH), near field measurements showed a higher particulate matter abundance and carbon dioxide (CO2) concentration as compared to far field measurements. We also found that aerosol viral load in far field is correlated with the number of particulates within the range of 1 µm -2.5 µm. Furthermore, increased ventilation and filtration are associated with lower environmental viral loads, and higher relative humidity is associated with lower aerosol viral loads and higher surface viral loads, consistent with an increased rate of particle deposition. Data from near field aerosol trials with high expiratory activities suggest that respiratory particles of smaller sizes (0.3 µm -1 µm) best characterize the variance of near field aerosol viral load. Moreover, our findings indicate that building operation practices such as ventilation, filtration, and humidification substantially reduce the environmental aerosol viral load, and therefore inhalation dose, and should be prioritized to improve building health and safety.
Summary by Key Questions:
- What is the relationship between human viral load and environmental contamination?
- We report a significant coefficient for all nasal and aerosol samples in routine trials whereby an increase in nasal viral load equivalent to -1 CTis associated with an increase in room aerosol viral load of -0.36216 CT(Supplemental figure 1). Furthermore, we report quantitative correlations between human viral load and high touched surfaces, as well as settling plates (Figure 3).
- Is there a difference in aerosol viral load at different distances from the infected participant, and is this affected by room air movement?
- We report that an increase in viral load equivalent to -1 CT in human nasal samples is significantly associated with an increased near field viral load of -0.32639 CT and an increased far field viral load of -0.4014 CT among routine trials (Figure 2a).
- The difference of means between the aerosol CT value of near field and far field aerosol samples in routine trials was 1.0583 CT, whereas far field samples represent lower viral load, however the paired t-test differentiating near field and far field samples was not significant (P = 0.05955). Interestingly, CO2concentration and the number of fine particles show statistically significant differences between near field and far field (Figure 2).
- How much ventilation do we need to significantly reduce aerosol viral load, and does this differ by distance from the infected individual?
- We observed that the aggregate of ventilation and filtration trials significantly reduced room aerosol viral load and that of select surfaces, when compared to control trials with ~0 ACH.
- When examining total room aerosol viral load (near field and far field together), we report that trials with less than ~4.5 ACH (including ~0 ACH trials) were associated with statistically higher viral load, by nearly an order of magnitude, than trials with greater than ~9 ACH (mean difference of -3.2 CT).We noticed that CO2has been frequently discussed among expert communities as an indicator of appropriate ventilation. Therefore, we provide the first real world correlation between aerosol viral load and CO2concentration that is affected by outdoor air exchange rates (figure 5b), where an increase in ~128 PPM of CO2concentration generated by an individual with COVID-19 corresponds with an increase in aerosol viral load equivalent to -1 CT, thus, approximately a doubling of the viral load.
- How much in-room filtration is needed to significantly reduce aerosol viral load, and does this differ by distance from the infected individual?
- We report that HEPA filtration trials (with ~1000 m3/hr) had significantly lower room aerosol viral load, by nearly an order of magnitude, when compared with control trials without filtration (mean difference of 3.240741 CT, Figure 5d).
- Does the level of indoor relative humidity significantly alter the aerosol viral load?
- We report that increasing relative humidity by ~11.85% is significantly associated with approximately an 50% decrease in aerosol viral load (Figure 6a).
- Which particle size bins best characterize the viral load?
- We investigated the correlation of particles and viral load through two different study set-ups (Figure 1) and found that aerosol viral load is significantly correlated with particles of smaller sizes (0.3 μm – 3μm) in near field and far field. In addition to fine particles, we observed statistically significant correlation between aerosol viral load and droplet size ranges (10 μm -25 μm) only during immediate field (< 1m) trials of high expiratory activity, but not during routine trials. For immediate field trials, particles of smaller sizes still best characterize the aerosol viral load.
Related BioBE COVID Research:
- March 2020 Building Mitigation Review Article – https://journals.asm.org/doi/10.1128/mSystems.00245-20 (published, mSystems)
- June 2020 SARS in HVAC – https://onlinelibrary.wiley.com/doi/10.1111/ina.12898 (published, Indoor Air)
- March 2021 Safe Air Spaces Risk Estimation – https://www.medrxiv.org/content/10.1101/2021.03.05.21252990v1 (Accepted, in press, Risk Analysis)
- March 2021 Modular Room Nebulization Study – https://www.preprints.org/manuscript/202103.0609/v1 (Accepted, in press, PlosOne)
- March 2021 Hospital Room Study – https://www.medrxiv.org/content/10.1101/2021.03.26.21254416v1
- August 2021 Dorm Room Study (includes effectiveness of natural ventilation) – https://www.researchsquare.com/article/rs-861942/v1 (In review)